Electroplating: Definition, Process, and Main Industrial Applications
What is Electroplating?
Electroplating is an electrochemical process that allows a metal object to be coated with a thin layer of another metal. This coating improves the aesthetic appearance of the part, its resistance to corrosion, durability and, when required, also certain functional properties such as hardness or electrical conductivity.
The operating principle is simple: the part is immersed in an electroplating tank containing a specific aqueous solution and two key elements, the anode and the cathode. By applying electric current, the metal ions present in the bath are transformed into solid metal and deposited onto the surface of the part.
In practice, electroplating is a controlled method to modify and improve the surface of a material in a precise and repeatable way.
It is widely used in many industrial sectors: fashion, accessories, metal hardware, faucets, automotive, electronics, medical, and many more.
Learn more about electroplating treatments offered by LEM.
How Electroplating Works
Although it is based on electrochemistry, the process can be explained with a few simple concepts.
Cathode, anode and electric current
During electroplating:
- the part to be treated is connected to the negative pole (cathode),
- another metal element (anode),
- an electric current flows through the tank.
When the part enters the solution, the circuit is closed. The positively charged metal ions in the bath are attracted to the cathode, gain electrons and turn into metal atoms that coat the part.
At the same time, oxidation reactions occur at the anode, releasing other species into the solution.
Why current density is fundamental
CEach electroplating bath operates within a precise current density range. Respecting this range is essential to obtain a uniform, defect-free deposit.
- Complex geometries (edges, cavities) receive different amounts of metal.
- If the current is too high, burning and irregular deposits appear.
- If it is too low, the metal appears dull and poorly levelled.
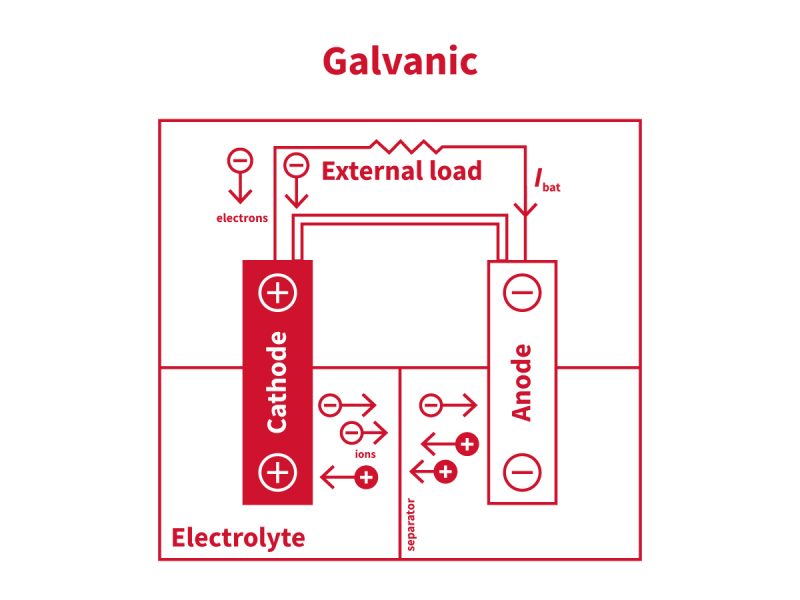
Chemical Fundamentals of Electroplating
Although each process has specific parameters, the main chemical components are common.
What an electroplating bath contains
Each bath consists of a mixture of elements working together:
- water
- metal salts
- organic additives that regulate brightness and leveling
- complexing agents that keep the metal stable in solution
- pH regulators
- surfactants, which improve wettability and reduce defects
The correct balance among these components determines the quality and stability of the process.
Deposit growth and typical defects
The metallic deposit grows through small crystalline nuclei that expand and merge.
The metal deposit grows from small nuclei that expand until they form a continuous layer.
If both the bath and the parameters are stable, the result is bright, uniform and adherent.
If the process is unbalanced, typical defects may appear:
- burning in high current density areas,
- inclusions and roughness due to particles or contaminants,
- poor leveling when additives are unbalanced,
- poor adhesion caused by insufficient pre-treatment,
- opalescence when organic components or additives are out of balance.
Many defects do not originate from the bath but from the raw part: oxides, machining residues or unsuitable materials.

The Importance of Pretreatments
Being conductive is not enough: the surface must be perfectly clean and activated before electroplating. For this reason, pre-treatments are a decisive phase for final quality.
The most common operations include:
Ultrasonic cleaning
Removes grease, polishing pastes and impurities thanks to the combined action of detergents and cavitation.
Electrolytic degreasing
Deeply cleans and activates the surface. Parts are connected to the cathode, and hydrogen production helps remove oxides and residues.
Neutralization
Eliminates alkaline residues left by the previous phase.
Pickling
Removes persistent oxides and prepares the substrate for deposition.
Without proper pre-treatment, the deposit may fail to adhere, appear stained or show visible defects.
Analytical Control of Electroplating Baths
Every bath changes over time: additives are consumed, metal content increases or decreases, and contaminants can enter the system. For this reason, scheduled controls are required, which may be daily, weekly or monthly.
Main analyses include:
- metal concentration measurement
- additive control through CVS or similar techniques
- verification of pH, conductivity and density
- analysis of metallic and organic contaminants
A well-structured analytical program helps prevent most defects.
An unbalanced bath generates problems that are difficult to correct. Monitoring ensures consistent quality, reduced scrap and longer bath life.
Types of Electroplating Plants
Plants differ by level of automation and the type of parts treated.
Manual systems
Ideal for metal fasteners, hardware and small components processed in large volumes. Visit LEM Manual electroplating plant.

Barrel plating
Suitable for small metal components, fasteners and bulk items. The rotating barrel allows parts to move inside the bath, ensuring uniform coating. Visit LEM Barrel electroplating plant.

Automatic lines
Ideal for high volumes and repetitive cycles. They allow precise control of all parameters and continuous process traceability. Visit LEM automatic lines.

Advantages, Limitations and Applications
Advantages
- high resistance to corrosion and wear
- wide variety of aesthetic finishes and colors
- improvement of surface mechanical properties
- possibility to deposit specific metals and alloys
Main limits
- need for constant bath monitoring
- rigorous pre-treatments
- precise environmental management
Applications
Fashion, faucets, electronics, automotive, furniture, medical devices and many others. Explore all LEM coating sectors.

Electroplating FAQ
Which parameters must be controlled in an electroplating bath?
Metal concentration, pH, temperature, conductivity, additive level and presence of contaminants.
How is bath stability maintained?
With periodic analyses, controlled replenishment, continuous filtration and temperature control.
Why are pre-treatments decisive?
Because deposit adhesion depends on the initial cleanliness and surface activation.
How is deposit quality evaluated?
Through non-destructive thickness measurements, adhesion tests, aesthetic evaluations and defect analysis.
Primary References
- G. Di Bari – Electrodeposition of Nickel, ASM International
- D.A. Jones – Principles and Prevention of Corrosion, Prentice Hall
- A.J. Bard, L.R. Faulkner – Electrochemical Methods, Wiley
- ASTM B456 – Standard Specification for Electrodeposited Coatings
- ISO 2080 – Electroplating and related processes – Vocabulary
Leonardo
Responsabile laboratorio chimico area galvanica - LEM srl
Did you find the article interesting? Share it now.
© 2026 Lem Galvanica. All rights reserved
Did you find something interesting? Contact us now!
LEM technicians are ready to address any questions or concerns you may have.
Request more information and discover our tailor-made solutions.
Other articles you may be interested in
StickyArticles
PVD vs. Electroplating: Differences, Advantages, and Limits
Discover the differences between PVD and electroplating: operating principles, resistance, costs, environmental impact, and a guide to choosing. Technical analysis by LEM.
Vibratory finishing: what it is and how it works
From degreasing to vibro-polishing, discover how LEM prepares metal surfaces. Focus on the Vintage finish for premium retro effects.
Sputtering: What It Is, How It Works, and PVD Applications
What is sputtering? Definition, plasma principle, process parameters, technical advantages, and main applications in PVD coatings.


